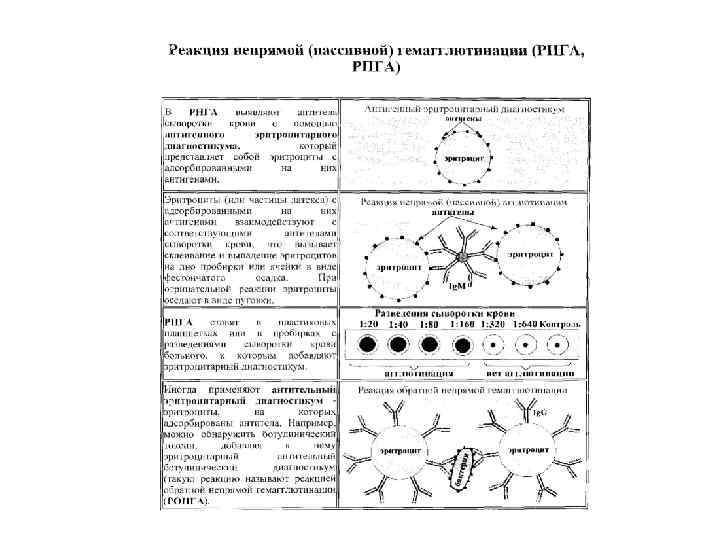
Mutation of the two charged residues at positions 49 Vk and 101 VH in HH8 to threonine precludes most of the salt links that are otherwise present in HH10 and HH26. HH8 can be seen to have the largest hydrophobic surface area, whereas HH26 has the largest charged-surface area. Molecular surface and intramolecular interactions of HH8 are shown in top-left and top-right panels, respectively those of HH10 are shown in middle-left and middle-right, respectively and those of HH26 are shown in bottom-left and bottom-right, respectively. The residues that participate in the intramolecular salt links in HH10 and HH26 (and those corresponding that are not involved in salt links in HH8) are shown in space-filling representation and the rest of the CDR residues in thin-ribbon representation. In the right panels, molecular figures depicting the presence and absence of intramolecular interactions (salt links) are shown. Surfaces corresponding to hydrophobic, polar, positively-charged, and negatively-charged residues are shown in brown, light blue, dark blue, and magenta, respectively. In the left panels, the CDR residues alone are shown in molecular surface representations. 
Figures prepared using QUANTA 97.Ĭomposite figure of surface representations and intramolecular salt links. HH10 and HH26 have their corresponding CDRs tightly packed due to the electrostatic interaction between their charged residues in L2 and H3. Additionally in HH26, E96 VH is also shown in green. K49 Vk and D101 VH residues of HH10 ( middle) and H26 ( bottom) are shown in magenta and green, respectively. HH8 ( top) has threonine at both the positions 49 Vk and 101 VH, shown in red and green, respectively (whereas they are lysines and aspartic acid, respectively, in both HH10 and HH26), and has hardly any interaction between its CDRs L2 and H3. Polar and hydrophobic residues of CDR L2 are shown in cyan and yellow, respectively, whereas most of the CDR H3 residues are in red. Heavy-chain, light-chain, and HEL are shown in pink, red, and blue, respectively. Stereo illustrations depicting the packing interactions between the residues of CDRs L2 and H3 in HH8 ( top), HH10 ( middle), and HH26 ( bottom), CDR residues alone shown in space-filling and rest of the atoms as thin-ribbon representations, respectively. Illustration of packing between CDRs L2 and H3. Results suggest that affinity maturation may select for high affinity antibodies with either "lock-and-key" preconfigured binding sites, or "preconfigured flexibility" by modulating combining site flexibility. The structurally rigid binding site with numerous specific contacts bestows specificity on HH26 whereas the flexible binding site with correspondingly fewer contacts enables HH8 to be cross-reactive. HH10 has these properties intermediate to HH8 and HH26. HH26 also has the most intermolecular contacts with the antigen whereas HH8 has the least. 
It is likely that the binding site of HH26 is rendered rigid by a network of intramolecular salt bridges whereas that of HH8 is flexible due to their absence. The modeled HH26-HEL structure predicts the recently determined x-ray structure of HH26, (Li et al., 2003, Nat. The binding site of HH26 is most charged, least hydrophobic, and has the greatest number of intramolecular salt bridges, whereas that of HH8 is the least charged, most hydrophobic and has the fewest intramolecular salt bridges. To investigate structural correlates of these functional variations, the Fv regions of HH8 and HH26 were homology-modeled using the x-ray structure of the well-characterized HH10-HEL complex as template. Their affinities vary by several orders of magnitude, and among the three antibodies, HH8 is the most cross-reactive with kinetics of binding that are relatively invariable compared to HH26, which is highly specific and has quite variable kinetics. Three antibodies, HyHEL-8 (HH8), HyHEL-10 (HH10), and HyHEL-26 (HH26) are specific for the same epitope on hen egg white lysozyme (HEL), and share >90% sequence homology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
